Katalüüsi uurimisrühm
Professor Tõnis Kangeri uurimisrühma laiem tegevusala on asümmeetriline orgaaniline süntees. Tegeldakse jätkusuutlike asümmeetrilise katalüüsi meetodite arendamise ja rakendamisega. Erinevaid katalüüsi meetodeid (organokatalüüsi, sh halogeensideme katalüüsi, metall- ja ensüümkatalüüsi) kasutatakse nii individuaalselt kui ka kombineeritult. Erilist tähelepanu pööratakse reaktsioonide efektiivsuse tõstmisele disainides ja kasutades selektiivseid multifunktsionaalseid katalüsaatoreid.
Uusi meetodeid rakendatakse looduslike ühendite ja nende derivaatide sünteesil, millest suurema praktilise väärtusega on rinnapiimas leiduvate oligosahhariidide (HMO-de) süntees.
Sünteetilist uurimistööd toetavad nii spektroskoopilised, kristallograafilised ja kromatograafilised eksperimendid kui ka teoreetilised kvantkeemilised arvutused. Läbiva teemana iseloomustab uurimistööd jätkusuutliku ja rohelise keemia printsiipide rakendamine asümmeetrilises sünteesis.
Tõnis Kanger on keemia ja biotehnoloogia instituudi orgaanilise sünteesi professor. Tema teaduslikud huvid hõlmavad katalüütilist asümmeetrilist sünteesi, aatomefektiivseid sünteetilisi meetodeid, multikomponentseid ja kaskaadreaktsioone ning asümmeetrilist organokatalüüsi. Ta on üle 100 artikli autor (h-indeks = 30, viidatud enam kui 2000 korda) ning tema teadustööd keemia ja molekulaarbioloogia alal on kahel korral tunnustatud Eesti Vabariigi teaduspreemiaga (2004, 2016).
CV link
tonis.kanger@taltech.ee
Researcher ID: H-2462-2015
ORCID: 0000-0001-5339-9682
Telefon: +372 620 4371
Faks: +372 620 2828
Tali, K.; Hunt, K. E.; Kriis, K.; Osadchuk, I.; Kanger, T. Anion-Assisted Glycosylation of Galactose: A Computational Study. J. Org. Chem. 2025, 90, 49, 17507–17517. DOI link
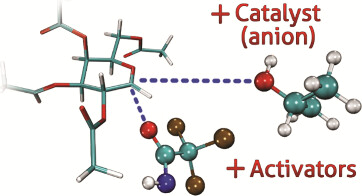
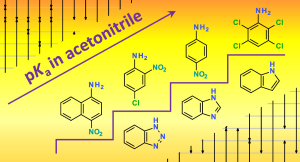
Lõkov, M.; Kesküla, C.; Tshepelevitsh, S.; Pikma, M.-L.; Saame, J.; Trubitsõn, D.; Kanger, T.; Leito,
I. The Acidity of Weak NH Acids: Expanding the pKa Scale in Acetonitrile. ACS Org. Inorg. Au 2025, 5, 2, 144–155. DOI link
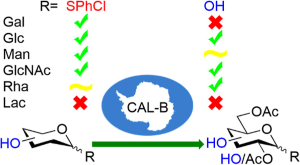
Hunt, K. E.; Miller, A.; Jarg, T.; Kriis, K.; Kanger, T. Selective Acetylation of Unprotected Thioglycosides and Fully Unprotected Monosaccharides with Candida antarctica Lipase-B. ACS Omega, 2025, 10, 19, 20047−20053. DOI link
Metsala, A.; Kriis, K.; Kanger, T. A DFT Study of an Organocatalytic Enantioselective Mannich Reaction Under the Sway of Noncovalent Interactions. Journal of Computational Chemistry, 2025, 46 (17), e70159. DOI link
Abstract: Density functional theory (DFT) calculations were performed for an asymmetric Mannich reaction between iminophosphorane and malononitrile catalyzed by a multifunctional organocatalyst. A quantum chemical modeling of the CC bond-forming reaction coordinate, proton transfer reaction coordinate, and torsional reaction coordinate was investigated with the help of M06-2X DFT calculations with a def2-SVP basis set. It was found that the rate-determining and enantiodetermining step (transition state) was neither associated with the CC bond-forming reaction coordinate nor with the proton transfer coordinate. The enantiodetermining step was associated with the torsional degrees of freedom and influenced by the network of non-covalent interactions.
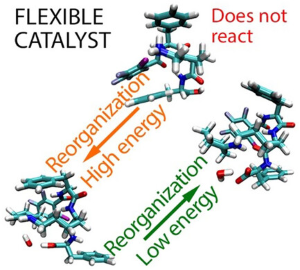
Osadchuk, I.; Kanger, T. Challenges in Mechanistic Investigation of a Flexible Aminocatalyst as Demonstrated through Enamine Formation. ChemistryOpen, 2025, 14 (9), e202500116. DOI link
Hunt, K. E.; Miller, A.; Liias, K.; Jarg, T.; Kriis, K.; Kanger, T. Interplay of Monosaccharide Configurations on the Deacetylation with Candida antarctica Lipase-B. J. Org. Chem., 2025, 90, 663−671. DOI link

Murre, A.; Erkman, K.; Kanger, T. Protocol for the preparation of primary amine-containing catalysts on the resin. STAR Protocols, 2024, 5, 1, 102933. DOI link

Murre, A.; Mikli, V.; Erkman, K.; Kanger, T. Primary amines as heterogeneous catalysts in an enantioselective [2,3]-Wittig rearrangement reaction. iScience, 2023 Sep 6; 26 (10):107822. DOI link

Sihtmae, M.; Silm, E.; Kriis, K.; Kahru, A.; Kanger, T. Aminocatalysts are More Environmentally Friendly than Hydrogen-Bonding Catalysts. ChemSusChem, 2022, 15, e202201045. DOI link

Hunt, K. E.; García-Sosa, A. T.; Shalima, T.; Maran, U.; Vilu, R.; Kanger, T. Org. Biomol. Chem., 2022, 20, 4724−4735. DOI link

Kriis, K.; Martõnov, H.; Miller, A.; Erkman, K.; Järving, I.; Kaasik, M.; Kanger, T. Multifunctional Catalysts in the Asymmetric Mannich Reaction of Malononitrile with N-Phosphinoylimines: Coactivation by Halogen Bonding versus Hydrogen Bonding. J. Org. Chem. 2022, 87 (11), 7422−7435. DOI link

Silm, E.; Järving, I.; Kanger, T. Asymmetric organocatalytic Michael addition of cyclopentane-1,2-dione to alkylidene oxindole. Beilstein Journal of Organic Chemistry 2022, 18, 167−173. DOI link

Murre, A.; Erkman, K.; Järving, I.; Kanger, T. Asymmetric Chemoenzymatic One-Pot Synthesis of α-Hydroxy Half-Esters. ACS Omega 2021, 6, 31, 20686–20698. DOI link

Kimm, M.; Järving, I.; Ošeka, M.; Kanger, T. Asymmetric Organocatalytic [2,3]‐Wittig Rearrangement of Cyclohexanone Derivatives. Eur. J. Org. Chem. 2021, 3113–3120. DOI link

Kaasik, M.; Martõnova, J.; Erkman, K,: Metsala, A.; Järving, I.; Kanger, T. Enantioselective Michael addition to vinyl phosphonates via hydrogen bond-enhanced halogen bond catalysis. Chem. Sci., 2021, 12, 7561-7568. DOI link

Trubitsõn, D.; Martõnova, J.; Kudrjašova, M.; Erkman; K.; Järving, I.; Kanger T. Enantioselective Organocatalytic Michael Addition to Unsaturated Indolyl Ketones. Organic Letters, 2021, 23 (5), 1820−1824. DOI link

Kõllo, M.; Kasari, M.; Kasari, V.; Pehk, T.; Järving, I.; Lopp, M.; Jõers, A.; Kanger, T. Designed whole-cell-catalysis-assisted synthesis of 9,11-secosterols. Beilstein Journal of Organic Chemistry 2021, 17, 581−588. DOI link
Abstract: A method for the synthesis of 9,11-secosteroids starting from the natural corticosteroid cortisol is described. There are two key steps in this approach, combining chemistry and synthetic biology. Stereo- and regioselective hydroxylation at C9 (steroid numbering) is carried out using whole-cell biocatalysis, followed by the chemical cleavage of the C–C bond of the vicinal diol. The two-step method features mild reaction conditions and completely excludes the use of toxic oxidants.
Trubitsõn, D.; Kanger, T. Enantioselective Catalytic Synthesis of N-alkylated Indoles. Symmetry 2020, 12, 1184. DOI link

Kaasik, M.; Kanger, T. Supramolecular Halogen Bonds in Asymmetric Catalysis. Frontiers in Chemistry 2020, 8, 599064. DOI link

Trubitsõn, D.; Martõnova, J.; Erkman, K.; Metsala, A.; Saame, J.; Kõster, K.; Järving, I.; Leito, I.; Kanger, T. Enantioselective N-Alkylation of Nitroindoles under Phase-Transfer Catalysis. Synthesis 2020, 52, 1047−1059. DOI link

Murre, A.; Erkman, K.; Kaabel, S.; Järving, I.; Kanger, T. Diastereoselective [2,3]-Sigmatropic Rearrangement of N-Allyl Ammonium Ylides. Synthesis 2019, 51, 4183−4197. DOI link

Silm, E.; Kaabel, S.; Järving, I.; Kanger, T. Asymmetric Organocatalytic Michael Addition–Cyclisation Cascade of Cyclopentane-1,2-dione with Alkylidene Malononitriles. Synthesis 2019, 51, 4198−4204. DOI link

Kimm, M.; Ošeka, M.; Kaabel, S.; Metsala, A.; Järving, I.; Kanger, T. [2,3]-Wittig Rearrangement as a Formal Asymmetric Alkylation of α-Branched Ketones. Organic Letters 2019, 21, 13, 4976-4980. DOI link

Kaasik, M.; Metsala, A.; Kaabel, S.; Kriis, K.; Järving, I.; Kanger, T. Halo-1,2,3-triazolium Salts as Halogen Bond Donors for the Activation of Imines in Dihydropyridinone Synthesis. Journal of Organic Chemistry 2019, 84, 4294−4303. DOI link

Kaasik, M.; Kaabel, S.; Kriis, K.; Järving, I.; Kanger, T. Synthesis of Chiral Triazole-Based Halogen Bond Donors. Synthesis 2019, 51, 2128-2135. DOI link

Peterson, A.; Kaasik, M.; Metsala, A.; Järving, I.; Adamson, J.; Kanger, T. Tunable chiral triazole-based halogen bond donors: assessment of donor strength in solution with nitrogen-containing acceptors. RSC Advances 2019, 9, 11718−11721. DOI link

Reitel, K.; Kriis, K.; Järvin, I.; Kanger, T. Study of the asymmetric organocatalyzed [3+2] annulation of cyclopropenone and β-keto ester. Chemistry of Heterocyclic Compounds 2018, 54, 929−933. DOI link

Trubitsõn, D.; Žari, S.; Kaabel, S.; Kudrjashova, M.; Kriis, K.; Järving, I.; Pehk, T.; Kanger, T. Asymmetric Organocatalytic Cascade Synthesis of Tetrahydrofuranyl Spirooxindoles. Synthesis 2018, 50, 314−322. DOI link

Ben Moussa, S.; Lachheb, J.; Gruselle, M.; Maaten, B.; Kriis, K.; Kanger, T.; Tõnsuaadu, K.; Badraoui, B. Calcium, Barium and Strontium apatites: A new generation of catalysts in the Biginelli reaction. Tetrahedron 2017, 73, 6542−6548. DOI link

Metsala, A.; Žari, S.; Kanger, T. Reaction path scans: Aza-Michael reactions of isatin imines. Computational and Theoretical Chemistry 2017, 1117, 30-40. DOI link

Kaasik, M.; Kaabel, S.; Kriis, K.; Järving, J.; Aav, R.; Rissanen, K.; Kanger, T. Synthesis and Characterisation of Chiral Triazole‐Based Halogen‐Bond Donors: Halogen Bonds in the Solid State and in Solution. Chemistry - A European Journal 2017, 23, 7337−7344. DOI link

Ošeka, M.; Kimm, M.; Järving, I.; Lippur, K.; Kanger, T. Two Catalytic Methods of an Asymmetric Wittig [2,3]-Rearrangement. J. Org. Chem. 2017, 82, 2889-2897. DOI link

Kriis, K.; Melnik, T.; Lips, K.; Juhanson, I.; Kaabel, S.; Järving, I.; Kanger, T. Asymmetric Synthesis of 2,3,4-Trisubstituted Piperidines. Synthesis 2017, 49, 604-614. DOI link

Metsala, A.; Žari, S.; Kanger, T. Aza-Michael Reactions of Isatin Imines: Deeper Insight and Origin of the Stereoselectivity. ChemCatChem 2016, 8, 2961-2967. DOI link

Ošeka, M.; Kimm, M.; Kaabel, S.; Järving, I.; Rissanen, K.; Kanger, T. Asymmetric Organocatalytic Wittig [2,3]-Rearrangement of Oxindoles. Org. Lett. 2016, 18, 1358-1361. DOI Link

Paju, A.; Kostomarova, D.; Matkevitš, K.; Laos, M.; Pehk, T.; Kanger, T.; Lopp, M. 3-Alkyl-1,2-cyclopentanediones by Negishi cross-coupling of a 3-bromo-1,2-cyclopentanedione silyl enol ether with alkylzinc reagents: An approach to 2-substituted carboxylic acid γ-lactones, homocitric and lycoperdic acids. Tetrahedron 2015, 71, 9313–9320. DOI Link

Preegel, G.; Ilmarinen, K.; Järving, I.; Kanger, T.; Pehk, T.; Lopp, M. Enantioselective Organocatalytic Michael Addition-Cyclization Cascade of Cyclopentane-1,2-dione with Substituted (E)-2-oxobut-3-enoates. Synthesis 2015, 47, 3805–3812. DOI Link

Lippur, K.; Kaabel, S.; Järving, I.; Rissanen, K.; Kanger, T. CaCl2, Bisoxazoline, and Malonate: A Protocol for an Asymmetric Michael Reaction. J. Org. Chem. 2015, 80, 6336-6341. DOI Link

Kaasik, M.; Noole, A.; Reitel, K.; Järving, I.; Kanger, T. Organocatalytic conjugate addition of cyclopropylacetaldehyde derivatives to nitro olefins: En route to β- and γ-amino acids. Eur. J. Org. Chem. 2015, 1745-1753. DOI Link

Žari, S.; Metsala, A.; Kudrjashova, M.; Kaabel, S.; Järving, I.; Kanger, T. Asymmetric organocatalytic aza-michael reactions of isatin derivatives. Synthesis 2015, 47, 875-886. DOI Link

Kreek, K.; Kriis, K.; Maaten, B.; Uibu, M.; Mere, A.; Kanger, T.; Koel, M. Organic and carbon aerogels containing rare-earth metals: Their properties and application as catalysts. J. Non-Cryst. Solids 2014, 404, 43-48. DOI Link

Maaten, B.; Moussa, J.; Desmarets, C.; Gredin, P.; Beaunier, P.; Kanger, T.; Tõnsuaadu, K.; Villemin, D.; Gruselle, M. Cu-modified hydroxy-apatite as catalyst for Glaser-Hay CC homo-coupling reaction of terminal alkynes. J. Mol. Catal. A 2014, 393, 112-116. DOI Link

Preegel, G.; Noole, A.; Ilmarinen, K.; Järving, I.; Kanger, T.; Pehk, T.; Lopp, M. Enantioselective Organocatalytic Michael Addition of Cyclopentane-1,2-diones to Nitroolefins. Synthesis 2014, 46, 2595–2600. DOI Link

Paju, A.; Kanger, T.; Müürisepp, A.-M.; Aid, T.; Pehk, T.; Lopp, M. Sonogashira cross-coupling of 3-bromo-1,2-diones: An access to 3-alkynyl-1,2-diones. Tetrahedron 2014, 70, 5843-5848. DOI Link

Žari, S.; Kudrjashova, M.; Pehk, T.; Lopp, M.; Kanger, T. Remote activation of the nucleophilicity of isatin. Org. Lett. 2014, 16, 1740-1743. DOI Link

Ošeka, M.; Noole, A.; Žari, S.; Öeren, M.; Järving, I.; Lopp, M.; Kanger, T. Asymmetric diastereoselective synthesis of spirocyclopropane derivatives of oxindole. Eur. J. Org. Chem. 2014, 3599-3606. DOI Link

Noole, A.; Malkov, A.; Kanger, T. Asymmetric organocatalytic synthesis of spiro-cyclopropaneoxindoles. Synthesis 2013, 45, 2520-2524. DOI Link

Noole, A.; Ilmarinen, K.; Järving, I.; Lopp, M.; Kanger, T. Asymmetric synthesis of congested spiro-cyclopentaneoxindoles via an organocatalytic cascade reaction. J. Org. Chem. 2013, 78, 8117-8122. DOI Link

Reitel, K.; Lippur, K.; Järving, I.; Kudrjašova, M.; Lopp, M.; Kanger, T. Asymmetric aminocatalytic Michael addition of cyclopropane-containing aldehydes to nitroalkenes. Synthesis 2013, 45, 2679-2683. DOI Link

Noole, A.; Ošeka, M.; Pehk, T.; Öeren, M.; Järving, I.; Elsegood, M.R.J.; Malkov, A.V.; Lopp, M.; Kanger, T. 3-Chlorooxindoles: Versatile starting materials for asymmetric organocatalytic synthesis of spirooxindoles. Adv. Synth. Catal. 2013, 355, 829-835. DOI Link

Ausmees, K.; Kriis, K.; Pehk, T.; Werner, F.; Järving, I.; Lopp, M.; Kanger, T. Diastereoselective multicomponent cascade reaction leading to [3.2.0]-heterobicyclic compounds. J. Org. Chem. 2012, 77, 10680-10687. DOI Link

Lippur, K.; Tiirik, T.; Kudrjashova, M.; Järving, I.; Lopp, M.; Kanger, T. Amination of quinolones with morpholine derivatives. Tetrahedron 2012, 68, 9550-9555. DOI Link

Noole, A.; Järving, I.; Werner, F.; Lopp, M.; Malkov, A.; Kanger, T. Organocatalytic asymmetric synthesis of 3-chlorooxindoles bearing adjacent quaternary-tertiary centers. Org. Lett. 2012, 14, 4922-4925. DOI Link

Žari, S.; Kailas, T.; Kudrjashova, M.; Öeren, M.; Järving, I.; Tamm, T.; Lopp, M.; Kanger, T. Organocatalytic asymmetric addition of malonates to unsaturated 1,4-diketones. Beil. J. Org. Chem. 2012, 8, 1452-1457. DOI Link

Reile, I.; Paju, A.; Kanger, T.; Järving, I.; Lopp, M. Cyclopentane-1,2-dione bis(tert-butyldimethylsilyl) enol ether in asymmetric organocatalytic Mukaiyama-Michael reactions. Tetrahedron Lett. 2012, 53, 1476-1478. DOI Link

Reinart-Okugbeni, R.; Ausmees, K.; Kriis, K.; Werner, F.; Rinken, A.; Kanger, T. Chemoenzymatic synthesis and evaluation of 3-azabicyclo[3.2.0]heptane derivatives as dopaminergic ligands. Eur. J. Med. Chem. 2012, 55, 255-261. DOI Link

Noole, A.; Pehk, T.; Järving, I.; Lopp, M.; Kanger, T. Organocatalytic asymmetric synthesis of trisubstituted pyrrolidines via a cascade reaction. Tetrahedron: Asymmetry 2012, 23, 188-198. DOI Link

Gruselle, M.; Kanger, T.; Thouvenot, R.; Flambard, A.; Kriis, K.; Mikli, V.; Traksmaa, R.; Maaten, B.; Tõnsuaadu, K. Calcium hydroxyapatites as efficient catalysts for the Michael C-C bond formation. ACS Catalysis 2011, 1, 1729-1733. DOI Link

Ausmees, K.; Selyutina, A.; Kütt, K.; Lippur, K.; Pehk, T.; Lopp, M.; Žusinaite, E.; Merits, A.; Kanger, T. Synthesis and biological activity of bimorpholine and its carbanucleosid. Nucleos Nucleot Nucl 2011, 30, 897-907. DOI Link

Noole, A.; Borissova, M.; Lopp, M.; Kanger, T. Enantioselective organocatalytic aza-ene-type domino reaction leading to 1,4-dihydropyridines. J. Org. Chem. 2011, 76, 1538-1545. DOI Link

Kriis, K.; Ausmees, K.; Pehk, T.; Lopp, M.; Kanger, T. A novel diastereoselective multicomponent cascade reaction. Org. Lett. 2010, 12, 2230-2233. DOI Link

Laars, M.; Raska, H.; Lopp, M.; Kanger, T. Cyclic amino acid salts as catalysts for the asymmetric Michael reaction. Tetrahedron: Asymmetry 2010, 21, 562-565. DOI Link

Noole, A.; Lippur, K.; Metsala, A.; Lopp, M.; Kanger, T. Enantioselective Henry reaction catalyzed by Cu(II) salt and bipiperidine. J. Org. Chem. 2010, 75, 1313-1316. DOI Link

Uudsemaa, M.; Kanger, T.; Lopp, M.; Tamm, T. pKa calculation for monoprotonated bipiperidine, bimorpholine and their derivatives in H2O and MeCN. Chem. Phys. Lett. 2010, 485, 83-86. DOI Link

Lippur, K.; Elmers, C.; Kailas, T.; Müürisepp, A.-M.; Pehk, T.; Kanger, T.; Lopp, M. Synthesis of 5,5'-disubstituted bimorpholines. Synth. Commun. 2010, 40, 266-281. DOI Link

Laars, M.; Ausmees, K.; Uudsemaa, M.; Tamm, T.; Kanger, T.; Lopp, M. Enantioselective organocatalytic Michael addition of aldehydes to β-nitrostyrenes. J. Org. Chem. 2009, 3772-3775. DOI Link

Uudsemaa, M.; Laars, M.; Kriis, K.; Tamm, T.; Lopp, M.; Kanger, T. Influence of protonation upon the conformations of bipiperidine, bimorpholine, and their derivatives. Chem Phys. Lett. 2009, 471, 92-96. DOI Link

Laars, M.; Kriis, K.; Kailas, T.; Müürisepp, A.-M.; Pehk, T.; Kanger, T.; Lopp, M. Structural constraints for C2-symmetric heterocyclic organocatalysts in asymmetric aldol reactions. Tetrahedron: Asymmetry 2008, 19, 641-645. DOI Link

Kanger, T.; Kriis, K.; Laars, M.; Kailas, T.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. Bimorpholine-mediated enantioselective intramolecular and intermolecular aldol condensation. J. Org. Chem. 2007, 72, 5168-5173. DOI Link

Kriis, K.; Laars, M.; Lippur, K.; Kanger, T. Bimorpholines as alternative organocatalysts in asymmetric aldol reactions. Chimia 2007, 61, 232-235. DOI Link
Abstract: Asymmetric organocatalytic aldol condensation is discussed on the basis of intramolecular and inter-molecular reactions. In addition to the widely used proline and its derivatives an application of the new type of the organocatalyst – bimorpholines in the above-mentioned reactions is described. The new catalyst has a unique C2-symmetric skeleton with four acceptor sites that makes it stereoselective and efficient. Small changes in the structure of the catalyst lead to a remarkable loss of selectivity.
Sulzer-Mossé, S.; Laars, M.; Kriis, K.; Kanger, T.; Alexakis, A. Synthesis and use of 3,3′-bimorpholine derivatives in asymmetric Michael addition and intramolecular aldol reaction. Synthesis 2007, 11, 1729-1732. DOI Link
Abstract: The synthesis of 3,3′-bimorpholine and its N-alkyl derivatives is described. These new diamine derivatives were revealed to be efficient organocatalysts for the asymmetric Michael addition of aldehydes to nitroalkenes with excellent enantioselectivity (up to 90% ee). The potential of these organocatalysts was also demonstrated for the highly enantioselective intramolecular aldol reaction affording the Wieland-Miescher ketone with tremendous enantioselectivity (up to 95% ee).
Aav, R.; Kanger, T.; Pehk, T.; Lopp, M. Synthesis of substituted cyclopentanones from 2-oxabicyclo[3.3.0]oct-6-en- 3-one. Proc. Est. Acad. Sci. Chem. 2007, 56, 3-13. Direct Link
Abstract: Synthesis of regioisomeric cyclopentanones 3-alkyloxy-2-oxabicyclo[3.3.0]octan-6-one (overall yield up to 34%) and 3-alkyloxy-2-oxabicyclo[3.3.0]octan-7-one (overall yield up to 18%) in four steps, starting from enantiomerically pure (–)-(1S,5R)-2-oxabicyclo[3.3.0]oct-6-en-3-one is described.
Lippur, K.; Kanger, T.; Kriis, K.; Kailas, T.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. Synthesis of (2S,2′S)-bimorpholine N,N′-quaternary salts as chiral phase transfer catalysts. Tetrahedron: Asymmetry 2007, 18, 137-141. DOI Link

Jõgi, A.; Paju, A.; Pehk, T.; Kailas, T.; Müürisepp, A.-M.; Kanger, T.; Lopp, M. Asymmetric synthesis of 2-aryl-5-oxotetrahydrofuran-2-carboxylic acids. Synthesis 2006, 3031-3036. DOI Link
Abstract: 3-Aryl-2-hydroxycyclopent-2-en-1-ones, when subjected to asymmetric oxidation, result in enantiomerically enriched 2-aryl-5-oxotetrahydrofuran-2-carboxylic acids. Electron-donating substituents in the para position of the phenyl ring increase the yield and decrease the enantioselectivity of the process.
Kriis, K.; Kanger, T.; Laars, M.; Kailas, T.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. Enantioselective synthesis of Wieland-Miescher ketone through bimorpholine-catalyzed organocatalytic aldol condensation. Synlett 2006, 1699-1702. DOI Link
Abstract: Novel bimorpholine-derived organocatalysts have been used for highly enantioselective intramolecular aldol reaction affording Wieland-Miescher ketone in high yield and enantioselectivity (up to 92% and 95%, respectively).
Paju, A.; Laos, M.; Jõgi, A.; Päri, M.; Jäälaid, R.; Pehk, T.; Kanger, T.; Lopp, M. Asymmetric synthesis of 2-alkyl-substituted 2-hydroxyglutaric acid γ-lactones. Tetrahedron Lett. 2006, 47, 4491-4493. DOI Link

Mossé, S.; Laars, M.; Kriis, K.; Kanger, T.; Alexakis, A. 3,3′-bimorpholine derivatives as a new class of organocatalysts for asymmetric Michael addition. Org. Lett. 2006, 8, 2559-2562. DOI Link

Kanger, T.; Laars, M.; Kriis, K.; Kailas, T.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. Anchimeric assistance in the case of vicinal dimesylate: Formation of enantiomeric or meso-bimorpholine. Synthesis 2006, 1853-1857. DOI Link
Abstract: An anchimeric effect of vicinal dimesylate in the intramolecular nucleophilic substitution by amine is described. One sulfonate group of the dimesylate acts as an internal nucleophile and the other as a leaving group, affording meso-bimorpholine in the intramolecular cyclization. ω,ω′-Dimesylate omits this effect and the target compound is obtained with high ee.
Kanger, T.; Raudla, K.; Aav, R.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. Synthesis and derivatization of bis-nor Wieland-Miescher ketone. Synthesis 2005, 3147-3151. DOI Link
Abstract: An efficient synthesis of bis-nor Wieland-Miescher ketone and its derivatives starting from commercially available 2-allyl-2-methylcyclopenta-1,3-dione is described.
Aav, R.; Kanger, T.; Pehk, T.; Lopp, M. Unexpected reactivity of ethyl 2-(diethylphosphono)propionate toward 2,2-disubstituted-1,3-cyclopentanediones. Phosphorus, Sulfur Silicon Relat. Elem. 2005, 180, 1739-1748. DOI Link
Abstract: A Horner-Wadsworth-Emmons reagent-ethyl 2-(diethylphosphono)propionate in the reaction with 2,2-disubstituted-1,3-cyclopentanediones, results in 4-oxohexanoic acid ethyl ester derivative up to 90% isolated yield. 31P-13C- and 1H-NMR study of the intermediates of the reaction involving the ethyl 2-(diethylphosphono)propionate was accomplished.
Paju, A.; Kanger, T.; Pehk, T.; Eek, M.; Lopp, M. A short enantioselective synthesis of homocitric acid-γ-lactone and 4-hydroxy-homocitric acid-γ-lactones. Tetrahedron 2004, 60, 9081-9084. DOI Link

Kriis, K.; Kanger, T.; Lopp, M. Asymmetric transfer hydrogenation of aromatic ketones by Rh(I)/bimorpholine complexes. Tetrahedron: Asymmetry 2004, 15, 2687-2691. DOI Link

Paju, A.; Kanger, T.; Niitsoo, O.; Pehk, T.; Müürisepp, A.-M.; Lopp, M. Asymmetric oxidation of 3-alkyl-1,2-cyclopentanediones. Part 3: Oxidative ring cleavage of 3-hydroxyethyl-1,2-cyclopentanediones: Synthesis of α-hydroxy-γ-lactone acids and spiro-γ-dilactones. Tetrahedron: Asymmetry 2003, 14, 2393-2399. DOI Link

Kriis, K.; Kanger, T.; Müürisepp, A.-M.; Lopp, M. C2-symmetric bimorpholines as chiral ligands in the asymmetric hydrogenation of ketones. Tetrahedron: Asymmetry 2003, 14, 2271-2275. DOI Link

Kanger, T.; Ausmees, K.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. A comparative study of the synthesis of C2-symmetric chiral 2,2′-biaziridinyls. Synlett 2003, 1055-1057. DOI Link
Abstract: Two comparative synthetic routes to new enantiomeric C2-symmetric Boc-protected biaziridinyls from tartaric ester were studied. Simplicity, high enantiomeric purity and high chemical yield of the target compound characterize the proposed methods. Also, unprotected biaziridinyl was synthesized and fully characterized.
Paju, A.; Kanger, T.; Pehk, T.; Lindmaa, R.; Müürisepp, A.-M.; Lopp, M. Asymmetric oxidation of 3-alkyl-1,2-cyclopentanediones. Part 2: Oxidative ring cleavage of 3-alkyl-1,2-cyclopentanediones: Synthesis of 2-alkyl-γ-lactone acids. Tetrahedron: Asymmetry 2003, 14, 1565-1573. DOI Link

Paju, A.; Kanger, T.; Pehk, T.; Müürisepp, A.-M.; Lopp, M. Asymmetric oxidation of 3-alkyl-1,2-cyclopentanediones. Part 1: 3-Hydroxylation of 3-alkyl-1,2-cyclopentanediones. Tetrahedron: Asymmetry 2002, 13, 2439-2448. DOI Link

Paju, A.; Kanger, T.; Pehk, T.; Lopp, M. Direct asymmetric α-hydroxylation of 2-hydroxymethyl ketones. Tetrahedron 2002, 58, 7321-7326. DOI Link

Kanger, T.; Kriis, K.; Pehk, T.; Müürisepp, A.-M.; Lopp, M. Asymmetric synthesis of novel C2-symmetric bimorpholines. Tetrahedron: Asymmetry 2002, 13, 857-865. DOI Link

Paju, A.; Kanger, T.; Pehk, T.; Lopp, M. Asymmetric oxidation of 1,2-cyclopentanediones. Tetrahedron Lett. 2000, 41, 6883-6887. DOI Link
Abstract: Cyclic 3-alkyl-1,2-cyclopentanediones undergo a direct asymmetric oxidation with the DET/Ti(OiPr)4/tBuOOH oxidative system, resulting in enantiomeric α-hydroxy compounds and ring-cleaved hydroxylated acids (lactones) up to 95% ee.
Aav, R.; Kanger, T.; Pehk, T.; Lopp, M. Synthesis of the AB-ring of 9,11-secosterols. Synlett 2000, 529-531. DOI Link
Abstract: The first total synthesis of AB-ring system of an antiproliferative and cytotoxic 9,11-secosterol 1 is described. Enantiomerically pure (3S,5S,6S,10S)-3,6-diacetoxy-10-methylbicyclo[4.4.0]decan-9-one 8 (steroidal numeration) was prepared from (S)-Wieland-Miescher ketone.
Alexakis, A.; Aujard, I.; Kanger, T.; Mangeney, P. (R,R)- and (S,S)-N,N'-dimethyl-1,2-diphenylethylene-1,2-diamine. Org. Synth. 1999, 76, 23-34. DOI Link

Rose-Munch, F.; Gagliardini, V.; Perrotey, A.; Tranchier, J.-P.; Rose, E.; Mangeney, P.; Alexakis, A.; Kanger, T.; Vaissermann, J. Two-step synthesis of homochiral monoaminals of tricarbonylphthalaldehydechromium complex. Chem. Commun. 1999, 2061-2062. DOI Link
Abstract: Tricarbonylphthalaldehydechromium complex can be prepared using a ‘one pot’ procedure starting from tricarbonylbenzenechromium: the protection of one aldehyde function by chiral diamines leads to the formation of two diastereoisomers of the monoaminal of the phthlaldehyde complexes, efficient precursors of enantiopure ortho-substituted alkenyl arene complexes.
Kanger, T.; Kriis, K.; Paju, A.; Pehk, T.; Lopp, M. Asymmetric oxidation of cyclobutanones: Modification of the sharpless catalyst. Tetrahedron: Asymmetry 1998, 9, 4475-4482. DOI Link
Abstract: Oxidation of prochiral and racemic cyclobutanones with t-BuOOH and Ti–TADDOL-based complexes afforded lactones in up to 44% ee. The enantioselectivity of the reaction clearly depends on the amount of the reagent and the highest enantioselectivities were obtained with stoichiometric amounts of the complex. Modification of the TADDOL structure and use of the mixed complex derived from TADDOL and a tartaric ester led to more reactive but less selective oxidation systems.
Kanger, T.; Niidas, P.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. Synthesis of chiral epoxyalkynes. Tetrahedron: Asymmetry 1998, 9, 2499-2508. DOI Link
Abstract: The synthesis of novel chiral propargylic epoxides ((R)-1-t-butyldimethylsilyl-3,4-epoxy-1-butyne, (3S,4S)-3,4-epoxy-1-octyne, (3R,4S)-1-t-butyldimethylsilyl-3,4-epoxy-1-pentyne) has been developed starting from the readily available tartaric acid derivative, (S,S)-(+)-2,3-O-isopropylidene-L-threitol.
Lopp, M.; Paju, A.; Kanger, T.; Pehk, T. Direct asymmetric α-hydroxylation of β-hydroxyketones. Tetrahedron Lett. 1997, 38, 5051-5054. DOI Link

Lopp, M.; Paju, A.; Kanger, T.; Pehk, T. Asymmetric Bayer-Villiger oxidation of cyclobutanones. Tetrahedron Lett. 1996, 37, 7583-7586. DOI Link

Kobzar, G.; Mardla, V.; Kanger, R.; Lopp, M.; Lille, U. Comparison of the Anti-Aggregatory activity of enantiomers of a 15-non-stereogenic carbacyclin analogue MIM706. Pharmacol. Toxicol. 1995, 76, 297-298. DOI Link
Alexakis, A.; Kanger, T.; Mangeney, P.; Rose-Munch, F.; Perrotey, A.; Rose, E. Enantioselective ortho-lithiation of benzaldehyde chromiumtricarbonyl complex. Tetrahedron: Asymmetry 1995, 6, 2135-2138. DOI link

Alexakis, A.; Kanger, T.; Mangeney, P.; Rose-Munch, F.; Perrotey, A.; Rose, E. Enantioselective ortho-Lithiation of Aminals of benzaldehyde chromiumtricarbonyl complex. Tetrahedron: Asymmetry 1995, 6, 47-50. DOI link

Kobzar, G.; Shelkovnikov, S.; Mardla, V.; Savitski, G.; Lopp, M.; Kanger, T.; Lille, U. A 15-nonstereogenic carbocyclic analogue of prostacyclin: Effects on human platelets and uterine artery. J. Lipid Mediat. Cell Sign. 1994, 10, 243-249. Link
Abstract: Platelet-suppressant and vasodilator effects of the (+) enantiomer of 13,14-dihydro-15,16,17,18,19,20-hexanor-14-(1-hydroxycyclohexyl)++ +carbocyclin (MM706), a carbocyclic analogue of prostacyclin with the 15-hydroxyl group located at a symmetrically substituted carbon atom were studied on human platelets and isolated uterine artery. In washed platelets it inhibits platelet aggregation with an IC50 value of 77 nM as compared to IC50 = 1.3 nM for PGI2. In the presence of indomethacin its potency like that of PGI2 decreases about 2-fold. Also, MM706 like PGI2 fails to inhibit the norepinephrine (NE)-stimulated contractions of isolated human uterine artery in the absence of indomethacin. Pretreatment of the artery with indomethacin (3 microM) makes it sensitive to both MM706 and PGI2, the EC50 values being 99 and 3.1 nM, respectively. The results show that MM706 is an effective inhibitor of platelet aggregation and NE-stimulated contractions of the indomethacin-treated uterine artery.
Kanger, T.; Liiv, M.; Pehk, T.; Lopp, M. A highly stereoselective synthesis of a new propargylic epoxide: (3R,4S)-1-tert-butyldimethylsilyl-3,4-epoxy-1-pentyne. Synthesis 1993, 91-93. DOI Link
Abstract: The synthesis of a novel enantiopure propargylic epoxide, (3R, 4S)-1-tert-butyldimethylsilyl-3,4-epoxy-1-pentyne (9), from a readily available tartaric acid derivative, (4R,5S)-5-{[(tert-butyldimethyl-silyl)oxy]methyl}-2,2-dimethyl-1, 3-dioxalane-4-carbaldehyde (1), is described.
Kanger, T.; Lopp, M.; Muraus, A.; Lohmus, M.; Kobzar, G.; Pehk, T.; Lille, U. Synthesis of a novel, optically active 15-nonstereogenic carbaprostacyclin. Synthesis 1992, 925-927. DOI Link
Abstract: Novel, optically active carbacyclin analogues (+)-1 and (-)-1 with an achiral ω-chain were synthesized from homochiral bromohydrins 3a and 4a, respectively.
Lopp, M.; Kanger, T.; Miiraus, A.; Pehka, T.; Lille, Ü. Synthesis of a novel four-carbon chiron - (R)-1-t-butyldimethylsilyl-3,4-epoxy-but-1-yne. Tetrahedron: Asymmetry 1991, 2, 943-944. DOI Link
Abstract: A simple and efficient synthesis of the novel chiron-(R)-1-Tl3DMS-3,4-epoxybut-1-yne has been developed starting from a derivative of (R,R)-(+)-tartaric acid. A new stereoselective bromination reaction of an O-silyl ether with BBr3 is also described.
Kanger, T.; Lopp.; Lille, Ü. Reactions of oxiranes. 2. Effect of protective groups on regioselectivity of oxirane opening in 2,3-epoxybicyclo[3.2.0]heptan-6-ones by lithium alkynide in the presence of boron-trifluoride. Zh. Org. Khim. 1991, 27, 1693-1700.
Kanger, T.; Kabat, M, Viha, E.; Lopp, M.; Lille, Ü. Optically-active synthons for synthesis of prostanoids. 1. Separation of 2-exo-bromine-3-endo-hydroxybicyclo[3.2.0]heptan-6-one enantiomers. Zh. Org. Khim. 1990, 26, 1611-1714.
Lopp, M.; Paju, A.; Kanger, T.; Välimäe, T.; Lille, Ü. Alkynylation of ethylene ketal of 1-chloro-4-bromo-1E-buten-3-one - synthesis of enyne and diene fragments of leukotriene and pheromones. Zh. Org. Khim. 1989, 25, 869-870.
Kanger, T.; Lopp, M.; Lille, Ü. Reactions of Oxiranes. 1. Role of Boron-Trifluoride in alkynation of bicyclic oxiranes. Zh. Org. Khim. 1988, 24, 2543-2546.
Pihl, L.; Kanger, T.; Talvik, A. Kinetic study of ionization of nitroalkanes in mixed solvents. 9. Phenylnitromethane and phenylnitromethane-d2 in aqueous dimethylsulfoxide and aqeous dimethylformamide. Org. React. 1984, 21, 436-440.
Jooksvad projektid
TARISTU24-TK13 "Keemilise sünteesi ja tehnoloogia teadustaristu", Tõnis Kanger, Tallinna Tehnikaülikool
TK228U1 "Strateegilise mineraalse ja süsiniku-põhise ressursi ringmajanduse tippkeskus", Riina Aav, Tallinna Tehnikaülikool
Lõppenud projektid
PRG1031 "Jätkusuutlike asümmeetrilise sünteesi meetodite arendamine ja rakendamine (1.01.2021−31.12.2025)", Tõnis Kanger, Tallinna Tehnikaülikool, Loodusteaduskond, Keemia ja biotehnoloogia instituut
SLTTI16143T "Molekulaarse Rakutehnoloogia Tippkeskus (TK143) (1.01.2016−1.03.2023)", Tanel Tenson, Tartu Ülikool, Loodus- ja täppisteaduste valdkond, tehnoloogiainstituut
PRG657 "Jätkusuutlike asümmeetrilise sünteesi meetodite arendamine ja rakendamine (1.01.2020−31.12.2020)", Tõnis Kanger, Tallinna Tehnikaülikool, Loodusteaduskond, Keemia ja biotehnoloogia instituut
PUT1468 "Halogeenside: kristallidest lahusesse (1.01.2017−31.12.2019)", Tõnis Kanger, Tallinna Tehnikaülikool, Loodusteaduskond, Keemia ja biotehnoloogia instituut
IUT19-32 "Suunatud asümmeetriline katalüütiline süntees: kompleksne integreeritud käsitlus (1.01.2014−31.12.2019)", Margus Lopp, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond, Tallinna Tehnikaülikool, Loodusteaduskond, Keemia ja biotehnoloogia instituut
TAR8103 "Keemilise bioloogia tippkeskus (28.03.2008−30.11.2015)", Margus Lopp, Tallinna Tehnikaülikool, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond, Keemiainstituut, Orgaanilise keemia õppetool
RLOTITIPP "Keemilise bioloogia tippkeskus (7.07.2008−31.08.2015)", Tanel Tenson, Tartu Ülikool, Tartu Ülikool, Loodus- ja tehnoloogiateaduskond, Tartu Ülikooli Tehnoloogiainstituut
VA433 "Hetrogeensete metall katalüsaatorite loomine apatiidi pinnal (1.07.2009−30.06.2014)", Kaia Tõnsuaadu, Tallinna Tehnikaülikool, Tallinna Tehnikaülikool, Keemia ja materjalitehnoloogia teaduskond, Anorgaaniliste materjalide teaduslaboratoorium
ETF8289 "Efektiivsus organokatalüüsis (1.01.2010−31.12.2013)", Tõnis Kanger, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond
SF0140060s12 "Katalüütilise asümmeetriline sünteesi ja stereokeemilise analüüsi meetodid ja rakendused (1.01.2012−31.12.2013)", Margus Lopp, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond
SF0142725s06 "Uued asümmeetrilise sünteesi strateegiad ja meetodid (1.01.2006−31.12.2011)", Margus Lopp, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond
ETF6662 "Kiraalsed diamiinid organokatalüütilistes ja metallkatalüütilistes asümmeetrilistes reaktsioonides (1.01.2006−31.12.2009)", Tõnis Kanger, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond
590L "Vähivastased sahhariidide ja nukleosiidide analoogid (1.09.2005−30.06.2009)", Margus Lopp, Tallinna Tehnikaülikool, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond, Keemiainstituut, Orgaanilise keemia õppetool
BF42 "Balticum Organicum Syntheticum 2006 (1.05.2005−1.10.2006)", Margus Lopp, Tallinna Tehnikaülikool, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond, Keemiainstituut, Orgaanilise keemia õppetool
G4976 "C2-sümmeetrilised heterotsüklilised kiraalsed mõjurid ja katalüsaatorite ligandid: süntees ja rakendused (1.01.2002−31.12.2005)", Tõnis Kanger, Tallinna Tehnikaülikool, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond, Keemiainstituut, Orgaanilise keemia õppetool
SF0351761s01 "Uued selektiivsed asümmeetrilise sünteesi meetodid ja nende kasutamine bioaktiivsete ühendite saamiseks (1.01.2001−31.12.2005)", Margus Lopp, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond
G3164 "Kiraalsed metallikompleksid asümmeetrilises sünteesis (1.01.1998−31.12.2001)", Tõnis Kanger, Tallinna Tehnikaülikool, Tallinna Tehnikaülikool, Matemaatika-loodusteaduskond, Keemiainstituut, Orgaanilise keemia õppetool

Dr. Kadri Kriis
vanemteadur

Dr. Kristin Erkman
vanemteadur

Dr. Kaarel Hunt
teadur

Harry Martõnov
doktorant-nooremteadur

Annette Miller
doktorant-nooremteadur
Kerli Tali
doktorant-nooremteadur
PhD tudengid
Aleksandra Murre, PhD 2023
Estelle Silm, PhD 2022
Dmitri Trubitsõn, PhD 2021
Mariliis Kimm, PhD 2021
Mikk Kaasik, PhD 2020
Kärt Reitel, PhD 2019
Maksim Ošeka, PhD 2017
Sergei Žari, PhD 2015
Artur Noole, PhD 2014
Kerti Ausmees, PhD 2013
Marju Laars, PhD 2010
Riina Aav, PhD 2005
MSc tudengid
Mia Peterson, MSc 2025
Maria Volokhova, MSc 2018
Ilona Juhanson, MSc 2017
Kristiina Lips, MSc 2016
Triin Melnik, MSc 2015
Birgit Maaten, MSc 2014
Anita Plado, MSc 2013
BSc tudengid
Ott Velmet, BSc 2025
Rudolf Ristkok, BSc 2025
Joel Pärnamets, BSc 2024
Karl Buntsev, BSc 2024
Allan Raudsepp, BSc 2023
Trine Kasemägi, BSc 2019
Mari-Liis Ludvig, BSc 2017
Aleksei Gvozdev, BSc 2015
Maarja Orle, BSc 2014
Kristel Jaiu, BSc 2013
Ahto Luuri, BSc 2012
Tõnis Tiirik, BSc 2012
Kristel Kütt, BSc 2011
Cindy Elmers, BSc 2009
Vjatšeslav Šuvalov, BSc 2008
Välistudengid
2019 Annette Miller (Regensburg Universität, Germany)
2017 Raminton Gnanagurunathan (University Pierre et Marie Curie, France)
2016 Linda Thaï (University Pierre et Marie Curie, France)
2013 Tobias Peter Michael Glesner (Technische Universität Darmstadt, Germany)
2012 Benjamin Schröder (Georg-August University Göttingen, Germany)
2011 Kadi Liis Saar (University of Cambridge, UK)
2009 Jean-Pierre Charles (Institut Universitaire de Technologie de Béthune, France)
2006 Céline Gregorski (Institut Universitaire de Technologie de Béthune, France)


















